A Community of Science Enthusiasts
We are an online resource designed to spark minds in the interest of science through appealing illustrations, complementary product guides, and interactive learning. From the basic to the more complex scientific principles – we offer a full hands-on experience! We encourage you to check out our expert advice on what laboratory equipment or scientific tool to use when performing experiments or collecting data.
Our Editorial Policy

Our Main Goal
We initially started this website to provide our work associates with a reliable science resource. Our goal is to make this a central location for the information dissemination of research-based material.

Our Expert Team
Our team is composed of semi-retired and active science faculty from all over the state. Each contribution is based on years of teaching experience, hypothesis-testing, and knowledge acquisition through training and continuing education. Together, we make sure that you get descriptive and factual information.

Our Product Reviews
You’ll find several up-to-date reviews on the tools and equipment we use. We determine which of the products are promising based on initial research and actual testing. The data we gather is then turned into an all-encompassing write-up.

Your Role
As a dedicated life-long learner, we encourage you to think. Our work is aimed to cultivate your curiosity when it comes to science learning. We are always open to collaboration and feedback. Let us know if there’s something we can change or improve.
Latest Posts

What’s the Best Textbook for Chemistry? Reviews & Guide
It can be unnerving to learn Chemistry, and the vast concepts make it difficult to study without a supplemental resource. If you are a professor

What’s the Best Hydrogen Water Bottle? Reviews & Guide
You can survive 3 weeks without food, but only 3 days without water. This scientific fact proves that a bottle of water is an essential
What’s the Best Model Kit For Organic Chemistry? Reviews Here
While textbooks are great supplemental resources, what better way to teach a student how Chemical bonds work than by giving them an organic Chemistry model

What’s the Best Scientific Calculator For Chemistry Subjects?
If Chemistry were easy, then a scientific calculator wouldn’t be of much use. Unfortunately, Chemistry is composed of complex calculations, and a reliable calculator is
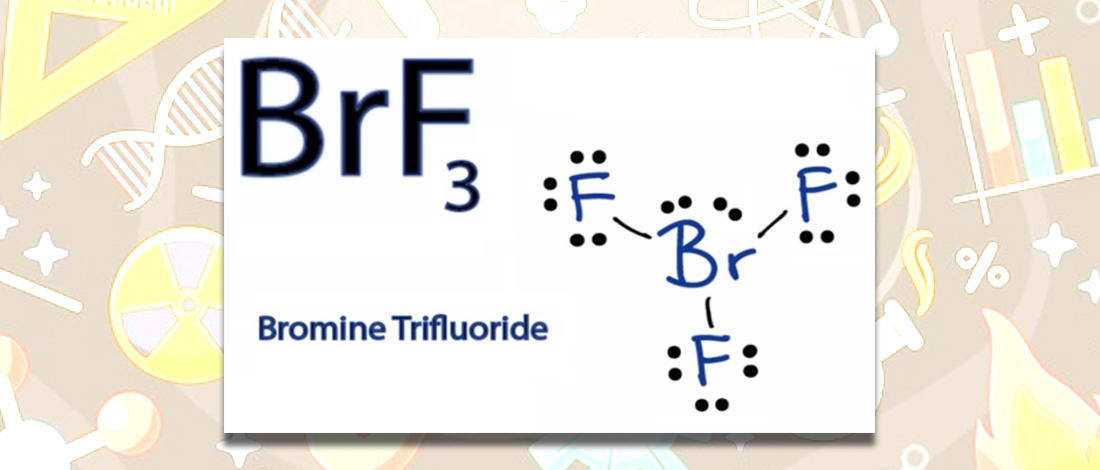
Molecular Geometry of Bromine Trifluoride
Bromine trifluoride is a strong interhalogen that is mainly used as a powerful fluorinating agent. This substance is commonly established in liquid form and has
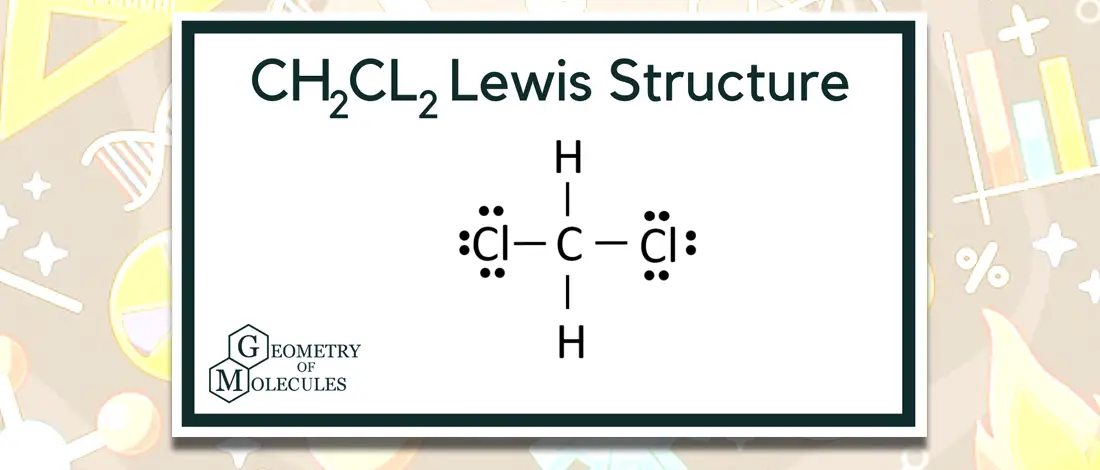
Is Dichloromethane Polar Or Nonpolar?
Dichloromethane, often known as methylene chloride, is an organic chemical compound. DCM’s chemical formula is CH2Cl2. It is an uncolored and volatile liquid with a
Most Popular Content

What’s the Best Textbook for Chemistry? Reviews & Guide
It can be unnerving to learn Chemistry, and the vast concepts make it difficult to study without a supplemental resource. If you are a professor
What’s the Best Textbook for a Biochemistry Subject?
Biochemistry textbooks tackle the common subfield of chemistry and biology that will help you study the subject. And when it comes to this, you can’t

What’s the Best Scientific Calculator For Chemistry Subjects?
If Chemistry were easy, then a scientific calculator wouldn’t be of much use. Unfortunately, Chemistry is composed of complex calculations, and a reliable calculator is

What’s the Best Model Kit For Organic Chemistry? Reviews Here
While textbooks are great supplemental resources, what better way to teach a student how Chemical bonds work than by giving them an organic Chemistry model

What’s the Best Hydrogen Water Bottle? Reviews & Guide
You can survive 3 weeks without food, but only 3 days without water. This scientific fact proves that a bottle of water is an essential

What’s the Best Digital USB Microscope? Reviews & Buying Guide
Microscopy is fun and educational, and with the right tools, there can be no limit to what someone can learn and discover. In this modern
As Seen On





